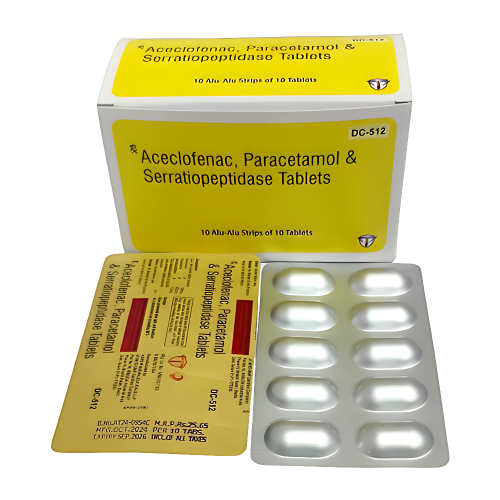
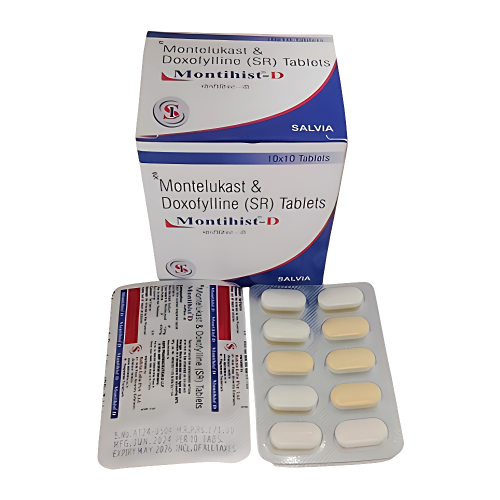
Aspo Pharmaceuticals
ASPO Pharmaceuticals LLP, a WHO-GLP-GMP Certified Manufacturing Unit
ASPO Pharmaceuticals LLP is a WHO-GMP and ISO 9001:2015 certified pharma medicine manufacturing company that specializes in manufacturing, research, development, export, and marketing. We offer a wide range of allopathic medicines as well as tablets, capsules, syrups, ointments. Our therapeutic specialties include cardiology, dermatology, paediatrics, gynaecology, critical care, and oncology.
ASPO Pharmaceuticals is an established manufacturer of pharmaceutical tablets in Himachal Pradesh (India). We serve over 600 B2B clients in more than six countries, providing high-quality pharmaceutical and nutraceutical solutions worldwide. Along with this, our ISO-approved pharma manufacturing unit is always known to focus on export and proudly possesses WHO-cGMP compliance and certification. This guarantees that every one of our products is of the highest quality and complies with international regulations.